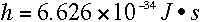Lab #17: Photoelectric Effect
4/1/2013
Lab Partner: Tara Miller
Sodium
C= λ.V h.V=E+W
λ=538nm, C=2.998E7 nm/s h=6.626E-34, E=0
V=(2.998E7)/(538)= 55724.91 s^-1 W=(6.626E-34)(55724.91 s^-1)= 3.692E-29
λ=538nm, C=2.998E7 nm/s h=6.626E-34, E=0
V=(2.998E7)/(538)= 55724.91 s^-1 W=(6.626E-34)(55724.91 s^-1)= 3.692E-29
Zinc
C= λ.V h.V=E+W
λ=287nm, C=2.998E7 nm/s h=6.626E-34, E=0
V=(2.998E7)/(287)= 1.045E15 s^-1 W=(6.626E-34)(1.045E15 s^-1)= 6.92E-19
λ=287nm, C=2.998E7 nm/s h=6.626E-34, E=0
V=(2.998E7)/(287)= 1.045E15 s^-1 W=(6.626E-34)(1.045E15 s^-1)= 6.92E-19
Copper
C= λ.V h.V=E+W
λ=262nm, C=2.998E7 nm/s h=6.626E-34, E=0
V=(2.998E7)/(262)= 114427.481 s^-1 W=(6.626E-34)(114427.481 s^-1)= 7.582E-29
λ=262nm, C=2.998E7 nm/s h=6.626E-34, E=0
V=(2.998E7)/(262)= 114427.481 s^-1 W=(6.626E-34)(114427.481 s^-1)= 7.582E-29
Questions
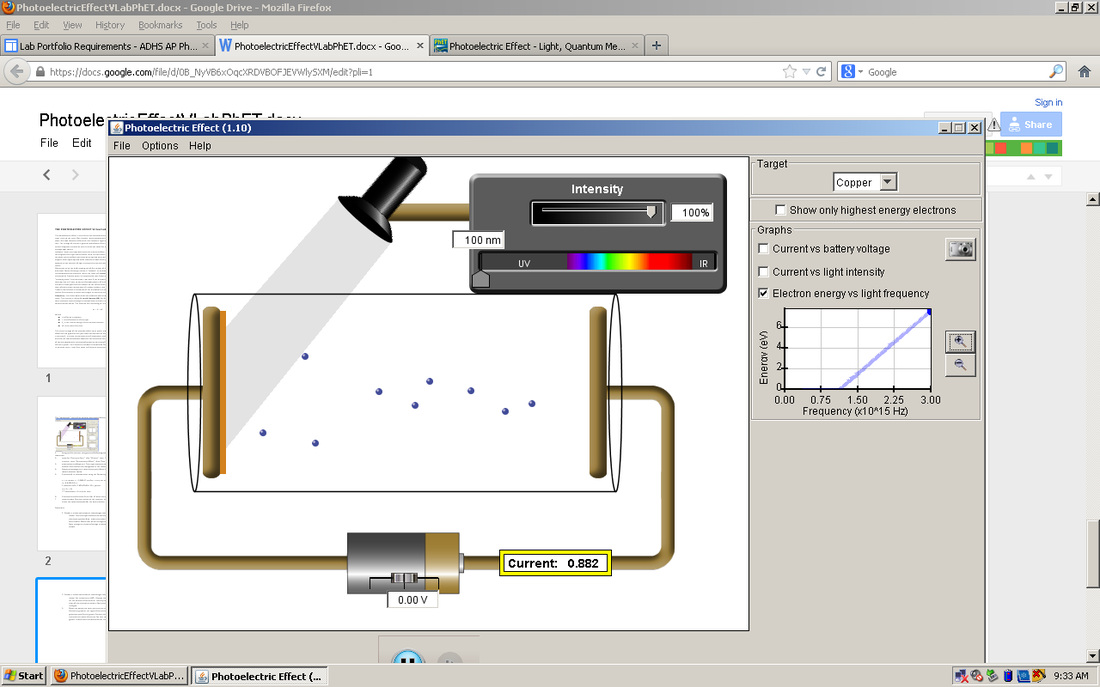
1.Metal: Copper
-λ=232: As intensity increases, the number of electrons increases.
More electrons get ejected as we decrease the wavelength while the intensity is constant; also, no electrons get ejected when the wavelength is greater than or equal to 263.E =nhf=hc/λ=eV: As wavelength decreases,the number of electrons that get ejected increases, and as intensity increases, the number of electrons increases.
2.Metal: Copper
λ=251nm: The flow of the electrons change because there is more power in the circuit and the particles speed up.
The number of charges also increases at both ends of the electron chamber.
3.Metal: Copper
λ=135 nm: The slope of the graph is the Planck's constant, which is:
-λ=232: As intensity increases, the number of electrons increases.
More electrons get ejected as we decrease the wavelength while the intensity is constant; also, no electrons get ejected when the wavelength is greater than or equal to 263.E =nhf=hc/λ=eV: As wavelength decreases,the number of electrons that get ejected increases, and as intensity increases, the number of electrons increases.
2.Metal: Copper
λ=251nm: The flow of the electrons change because there is more power in the circuit and the particles speed up.
The number of charges also increases at both ends of the electron chamber.
3.Metal: Copper
λ=135 nm: The slope of the graph is the Planck's constant, which is:
We obtained this value as the slope because E = nhf; h (Planck's constant) is the slope. E is energy, and f is the light frequency.
Reflection
This lab helped me to grasp the formulas and the Planck's constant better and understand the relationship between wavelength, intensity, and the number of electrons.